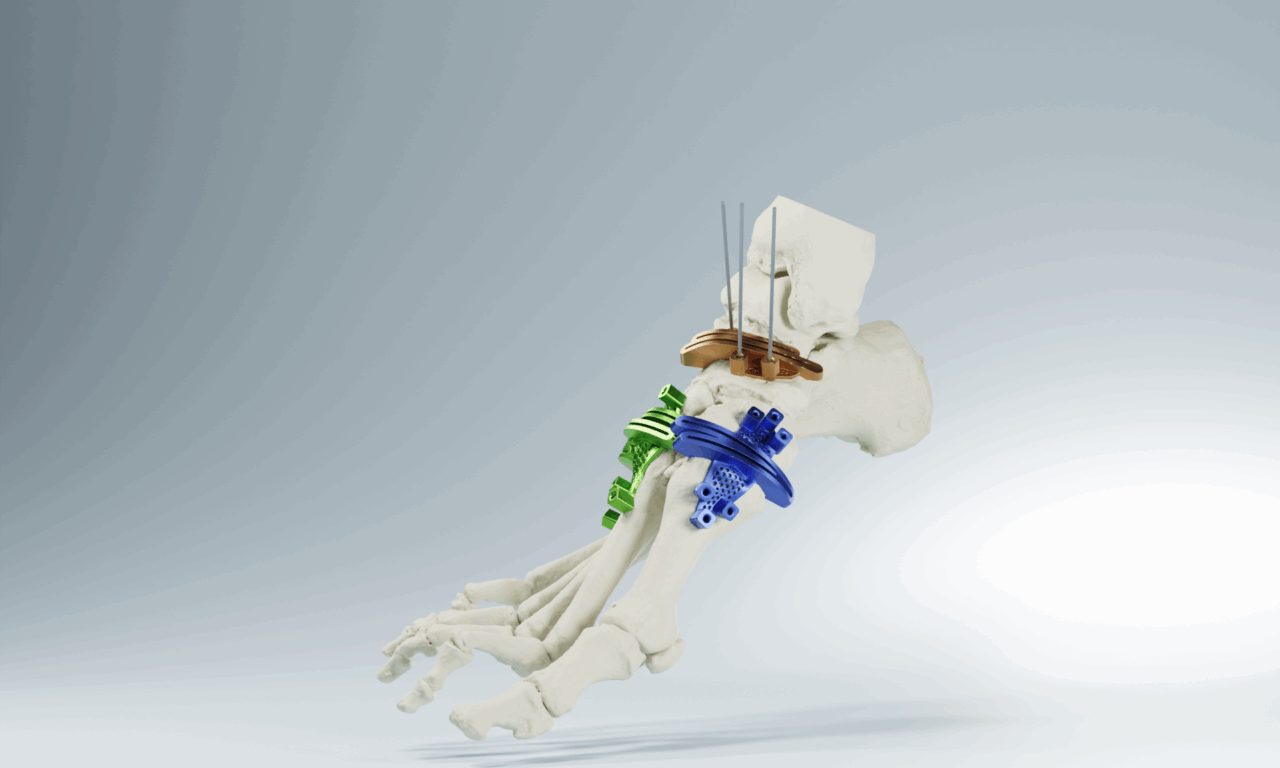
AccuStride™ Foot + Ankle System
Lower Extremity Reconstruction
Overview
The AccuStride™ System delivers patent-pending patient-specific surgical guides, plating, and anatomical models designed to streamline complex foot procedures. Using detailed CT imaging and surgeon collaboration, each case is tailored to support precise correction, stable fixation, and procedural efficiency. Indicated for use in trauma, general surgery, and reconstructive procedures of the foot, AccuStride™ enables a highly accurate, truly custom approach to diverse clinical challenges. AccuStride™ Plates and Guides are manufactured in titanium alloy to achieve an optimized anatomical fit.